SOLVED: For a gas at a given temperature, the compression factor is described by the empirical equation: z = 1 - 8.50 × 10^(-3)P/P° + 3.50 × 10^(-5)(P/P°)^2 where P° = 1
4.9 (313) In stock

VIDEO ANSWER: Hello students: let's look at the question: l n, that integrate integration and 0 z minus 1 bracket, close d p by p here. Minus 1 is equal to minus 8.50 into 10 to the power minus 3 p by p, not plus 3.50 into 10. To the power minus 9. P
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.

Federal Register :: Medicare and Medicaid Programs; CY 2023 Payment Policies Under the Physician Fee Schedule and Other Changes to Part B Payment and Coverage Policies; Medicare Shared Savings Program Requirements; Implementing

Schaum's Outlines - 3,000 Solved Problems in Chemistry, PDF, Chemical Bond


Engineering Thermodynamics35M, PDF, Internal Combustion Engine

Compression Factor and Fugacity

PDF) CHAPTER 1 CHEMISTRY: THE STUDY OF CHANGE Problem Categories

PDF) Seismic Fragility of Buried Steel Natural Gas Pipelines due to Axial Compression at Geotechnical Discontinuities
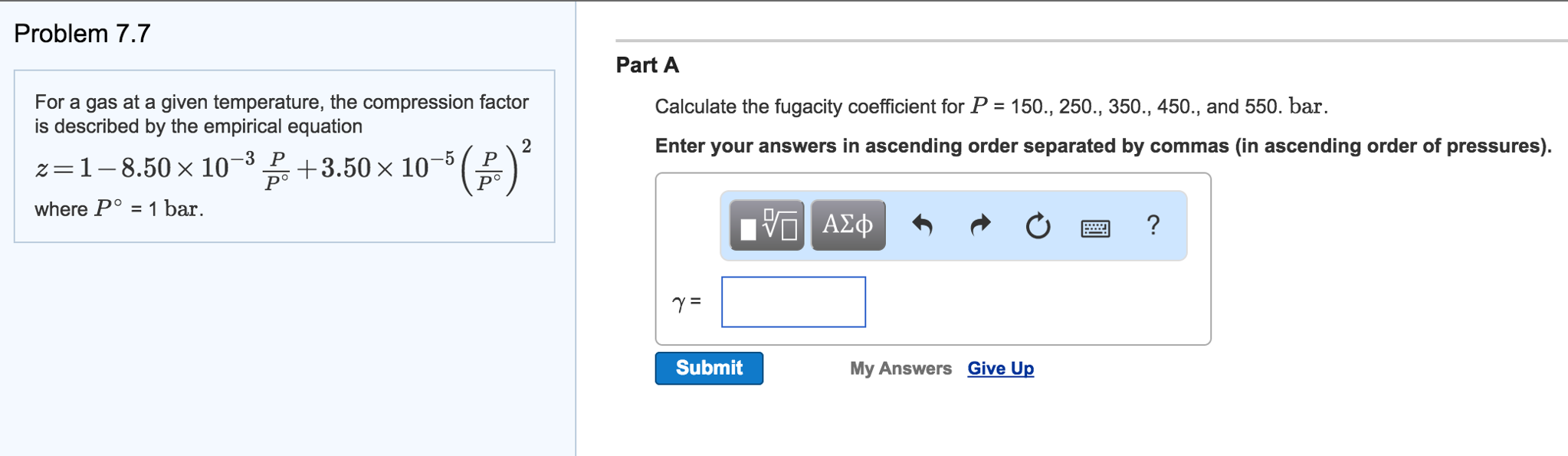
For a gas at a given temperature, the compression

At a given temperature T, gases Ne, Ar, Xe and Kr are found to deviate from ideal gas behaviour. Their equation of state is given as p = dfrac {RT}{V - b}

SOLVED: For a gas at a given temperature, the compression factor is described by the empirical equation: z = 1 - 8.50 × 10^(-3)P/P° + 3.50 × 10^(-5)(P/P°)^2 where P° = 1

Development of equations describing transport of mass, momentum and energy in soils

Solved ER4 - P7.13 (10 pts) For a gas at a given
COMPRESSION AND EXPANSION OF GASES – Chemical Engineering Projects





