Compressibility factor (Z) for a van der Waals real gas at critical point is
4.7 (436) In stock

Share your videos with friends, family and the world

Calculate the compressibility factor (z) for a Vander Waals gas at critical point? - EduRev NEET Question

COMPREHENSION_TYPE from IIT-JEE PREVIOUS YEAR (CHEMISTRY) STATES OF MATTER for Class 12

Compressibility factor (Z) for a van der Waals real gas at critical po

Solved Problem 2. ( 30 points) The ideal gas law can
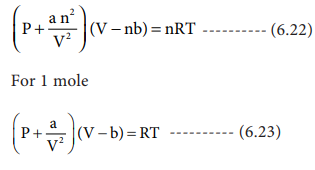
Derivation of critical constants from van der Waals constant
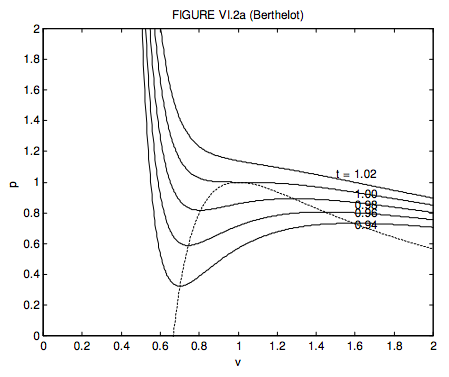
6.3: Van der Waals and Other Gases - Physics LibreTexts

Solved Problem 1. 15 points Find the compressibility factor

PDF) A Modified Form of the van der Waals Equation of State

Under critical conditions, the compressibility factor for a gas is .

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as
Compressibility Factor from Redlick-Kwong Equations
Compressibility factor (gases) - Knowino
Explain how the compression factor varies with pressure and
 Me - Women's Cotton Spandex Legging – Twisted Gear, Inc.
Me - Women's Cotton Spandex Legging – Twisted Gear, Inc. Brandon Maxwell Pre-Spring 2023 Resort - RUNWAY MAGAZINE ® Official
Brandon Maxwell Pre-Spring 2023 Resort - RUNWAY MAGAZINE ® Official Ways of Addressing Breast Asymmetry - Plastic Surgery Center of Dallas
Ways of Addressing Breast Asymmetry - Plastic Surgery Center of Dallas Stay stylish with essential workout pieces.
Stay stylish with essential workout pieces. Black Solid Polyester Spandex Women Slim Fit Tights - Selling Fast at
Black Solid Polyester Spandex Women Slim Fit Tights - Selling Fast at koi Next Gen On the Move Women's 8-Pocket Maternity Scrub Pants – koihappiness
koi Next Gen On the Move Women's 8-Pocket Maternity Scrub Pants – koihappiness