42g of N₂ react with excess of O₂ to produce NO. Amount of NO formed is a.60g b.32g c.45g d.90g
4.9 (122) In stock

Share your videos with friends, family and the world

Solved Nitrogen and oxygen react to produce nitric oxide

WO2013122236A1 - Base-protected oligonucleotide - Google Patents

Solved Nitrogen and oxygen react as follows: N2(g) +202(g) +

Mole Concept PDF, PDF, Mole (Unit)

Empirical formula of a hydrocarbon having 80% C and 20% of

Consider the reaction 2 NO + O2 $ o$ 2 NO2 Suppose that at

Solved If 42.0 g of nitrogen gas reacts with an excess of

Solved Use the balanced equation for the reaction of N2 and
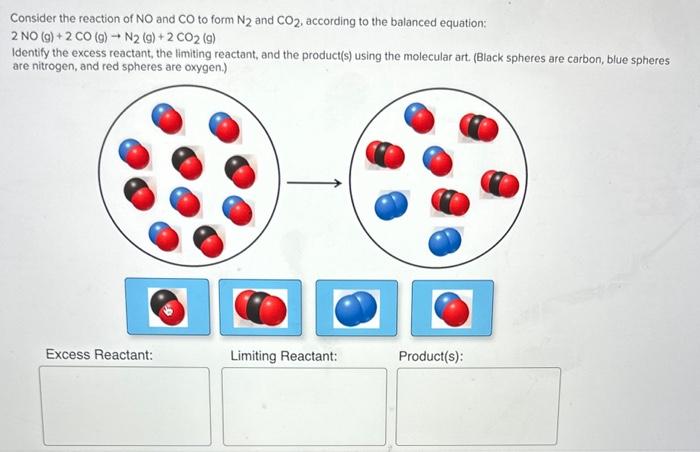
Solved Consider the reaction of NO and CO to form N2 and

WO2022045231A1 - Ester compound - Google Patents


Topical Mock Chemistry Questions, PDF, Chemical Elements
Kit 2 Caixas Chocolate Snickers Dark Barra Recheada C/20 42g
A&D Weighing HR-202-C Orion Analytical Balance, 42g/210g from Cole
Ottogi Spicy Vegetable Soup 21g x 2 Packs, 42g - H Mart Manhattan Delivery





