Derive an expression for the compression factor of a gas tha
4.8 (142) In stock
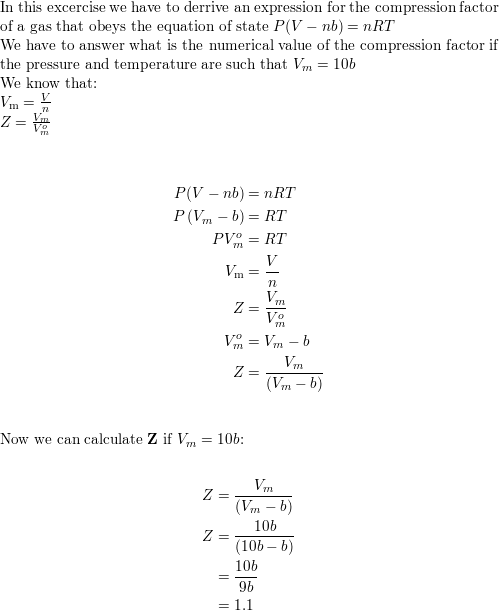
Equations of Compressible and Incompressible Flow in Fluid Dynamics, System Analysis Blog

SOLVED: Devise an expression for the compression factor of a gas that obeys the equation PVm - b = RT, where b and R are constants. If the pressure and temperature are

18 Tugas Sifat Sifat Gas CH 1, PDF, Gases

Isentrophic Compression, Glenn Research Center
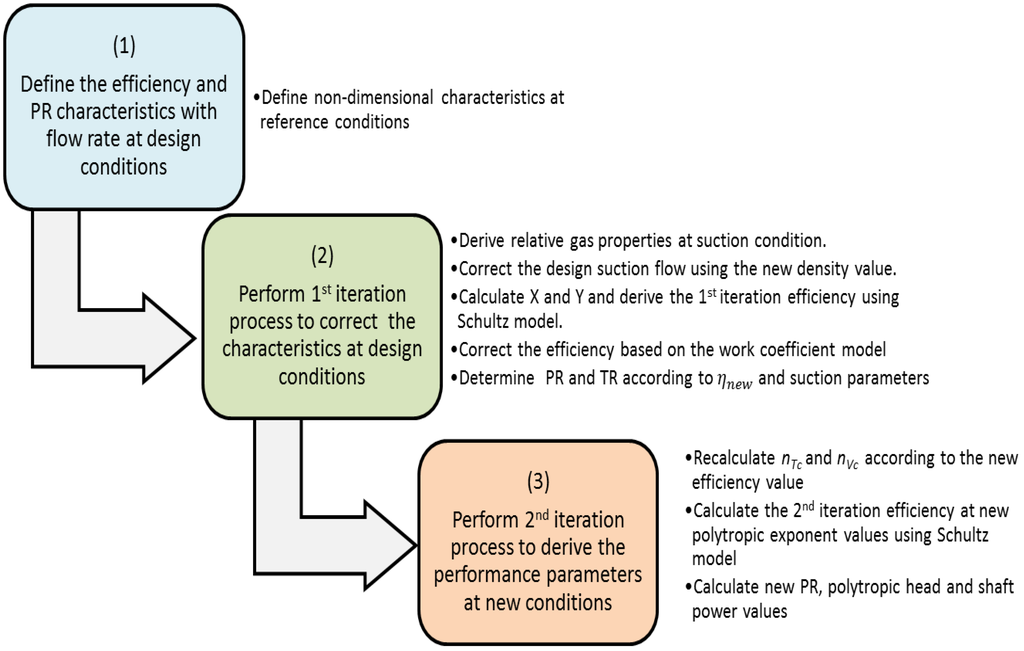
Energies, Free Full-Text

SOLVED: Devise an expression for the compression factor of a gas that obeys the equation PVm - b = RT, where b and R are constants. If the pressure and temperature are

SOLVED: The Berthelot equation of state is given by p = nRT/(V-nb). The total differential of the compression factor Z = PV is given by dZ = -MdT + NdV. (a) Write

Solved 8. Use the Maxwell distribution of speeds to estimate

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks
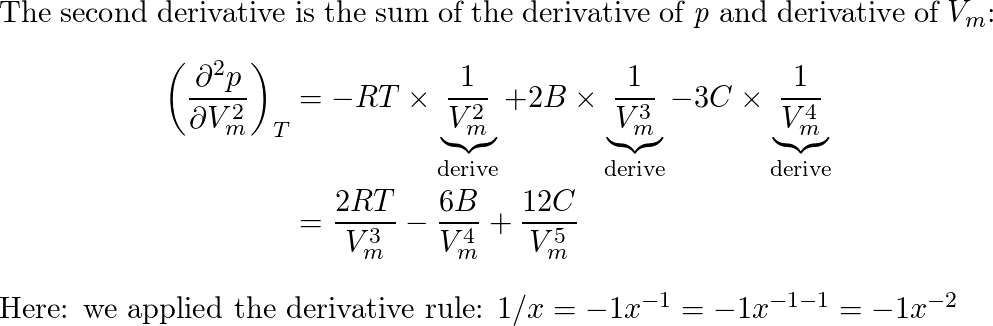
A scientist proposed the following equation of state $p= ra
The value of compression factor at the critical state of a vander waals gas is
Solved As a first approximation, the compression factor, Z
Solved 1. The compression factor, Z of a gas is 0.625. Which
COMPRESSION AND EXPANSION OF GASES – Chemical Engineering Projects
Solved Z = 4. We saw in class that the compression factor
Find the isothermal compressibility `x` of a Van der Walls gas as a function of volume
 Tommy Hilfiger Men's Burnished Handlaced Braided Belt (Pack of 2), 36, Black and Tan
Tommy Hilfiger Men's Burnished Handlaced Braided Belt (Pack of 2), 36, Black and Tan Blush Pink™ Nandina - Southern Living Plants
Blush Pink™ Nandina - Southern Living Plants News: Slap Bracelet MagicBands Are BACK in Disney World and Online!
News: Slap Bracelet MagicBands Are BACK in Disney World and Online! Kohl's Maidenform ® One Fabulous Fit® Underwire T-Shirt Bra DM7543 48.00
Kohl's Maidenform ® One Fabulous Fit® Underwire T-Shirt Bra DM7543 48.00 How to Smell Sweet Without Chemical Perfume
How to Smell Sweet Without Chemical Perfume MIER Men's 2 in 1 Running Shorts with Liner 5 Quick Dry Gym Workout Athletic Shorts with Pockets, Lightweight, Breathable : : Clothing
MIER Men's 2 in 1 Running Shorts with Liner 5 Quick Dry Gym Workout Athletic Shorts with Pockets, Lightweight, Breathable : : Clothing