117. Compressibility factor H, behaving as rea gas is 1) 1 RTV 3) 1+- RT 4) (1-a) 18. If V is the observed molor unlum
4.6 (93) In stock

Click here:point_up_2:to get an answer to your question :writing_hand:117 compressibility factor for h behaving as reagas is1 1rtv31rt41a18 if v is the observed
Click here👆to get an answer to your question ✍️ 117- Compressibility factor H- behaving as rea gas is 1- 1 RTV 3- 1- RT 4- -1-a- 18- If V is the observed molor unlum

The compressibility factor a real gas high pressure is:1+ dfrac{RT}{pb}1+ dfrac{pb}{RT}11- dfrac{pb}{RT}

XtraEdge for IIT-JEE 1 JUNE 2010 - Career Point

Real Gases Introductory Chemistry

3.2 Real gas and compressibility factor – Introduction to Engineering Thermodynamics

For a gas having value of compressibility factor greater than one, the volume of 1 mole of gas, Vmat S.T.P is e answer: Vm= 22.4L V> 22.4L Vm = 22.4L
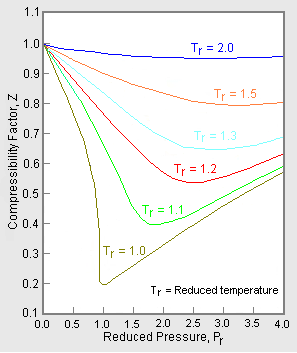
Compressibility factor (gases) - Citizendium

At low pressure, the compressibility factor of one mole of gas can be written as Brand pramu mb) sono (1) 1-9 271 RTV (3) 1-RT (4) 1- av hinh of the following has maximum numbertof F

Solved Use the compressibility charts to answer the

Welcome to Chem Zipper.com: The compressibility factor for 1 mole of a van der Waals gas at 0oC and 100 atm pressure is found to be 0.5. Assuming that the volume of

Foam FLOW-3D

a) A gas at 250 K and 15 atm has a molar volume 12 per cent

The graph of compressibility factor (Z) v/s P 1 mol of a real gas is shown in following diagram. The graph is plotted 273 K temperature. If slope of graph very high

171. CH4 gas is behaving non-ideally. Compressibility factor gas is 1.5 2 atm, 400 K. Calculate molar volume gas: [Given : R=0.08 Litre-atm, K-mole (1) 24 litre (2) 16 litre (3) 48 litre (4) 8 litre

The compression factor (compressibility factor) for `1 mol` of a van der Waals gas at
Ideal Gas Equation and COMPRESSIBILITY Factor in 11 Minutes!
Explain how the compression factor varies with pressure and
Compression Factor Calculator - Calculator Academy
the equation of state of a gas is p(v-nb)=rt where b and r are consta - askIITians





